Formic acid (HCOOH) is the simplest organic acid, naturally occurring in ant stings and known for its potent, corrosive properties in industrial use. Because of its strength, many people, especially those searching for “is formic acid a strong acid,” mistakenly assume it belongs in the category of strong mineral acids.
This article will meticulously clarify the chemical properties of formic acid, detail the definitive criteria used to distinguish between strong and weak acids, and compare its strength and application to familiar counterparts, including acetic acid, propionic acid, and benzoic acid.
The Chemistry of Formic Acid: Defining Acid Strength
Formic acid is classified as a carboxylic acid (R-COOH). Its unique structure—a single hydrogen atom attached directly to the carboxyl group—is key to its surprising power.
How Do Chemists Define Strong vs. Weak Acids?
The strength of any acid is quantified by its tendency to donate a proton (H+) in water, which is precisely measured by the pKa value (the negative logarithm of the acid dissociation constant, Ka).
- Strong Acids: These acids dissociate completely (100% ionization) in water, meaning every molecule releases an H+ ion. Their pKa values are typically very low, often negative (e.g., HCl).
- Weak Acids: These acids dissociate partially in water, establishing an equilibrium where most molecules remain intact. Their pKa values are higher.
The measured pKa value for formic acid is approximately 3.75. Since this is well above the threshold for strong acids (which is generally pKa < 2), formic acid is scientifically classified as a weak acid.
Common Strong Acids and Their Applications
Strong acids are essential in demanding industrial processes due to their ability to provide high concentrations of reactive H+ ions.
| Strong Acid | pKa (Approx.) | Primary Industrial Use |
|---|---|---|
| Hydrochloric Acid (HCl) | -6.3 | Metal surface cleaning (pickling), and chemical synthesis. |
| Sulfuric Acid (H2SO4) | -3.0 | The largest volume industrial chemical, critical for fertilizer production. |
| Nitric Acid (HNO3) | -1.4 | Manufacturing fertilizers, dyes, and explosives. |
Applications of Formic Acid: Leveraging a Powerful Weak Acid
Formic acid’s intermediate strength—potent enough for demanding tasks, yet milder than mineral acids—makes it the preferred organic acid for specific applications:
- Preservation and Disinfection: Used as a silage and feed preservative to inhibit bacterial and fungal growth, thanks to its higher acidity.
- Textile and Leather: Serves as a reducing agent in dyeing and finishing processes.
- Beekeeping: Used to control mites that threaten honeybee colonies.
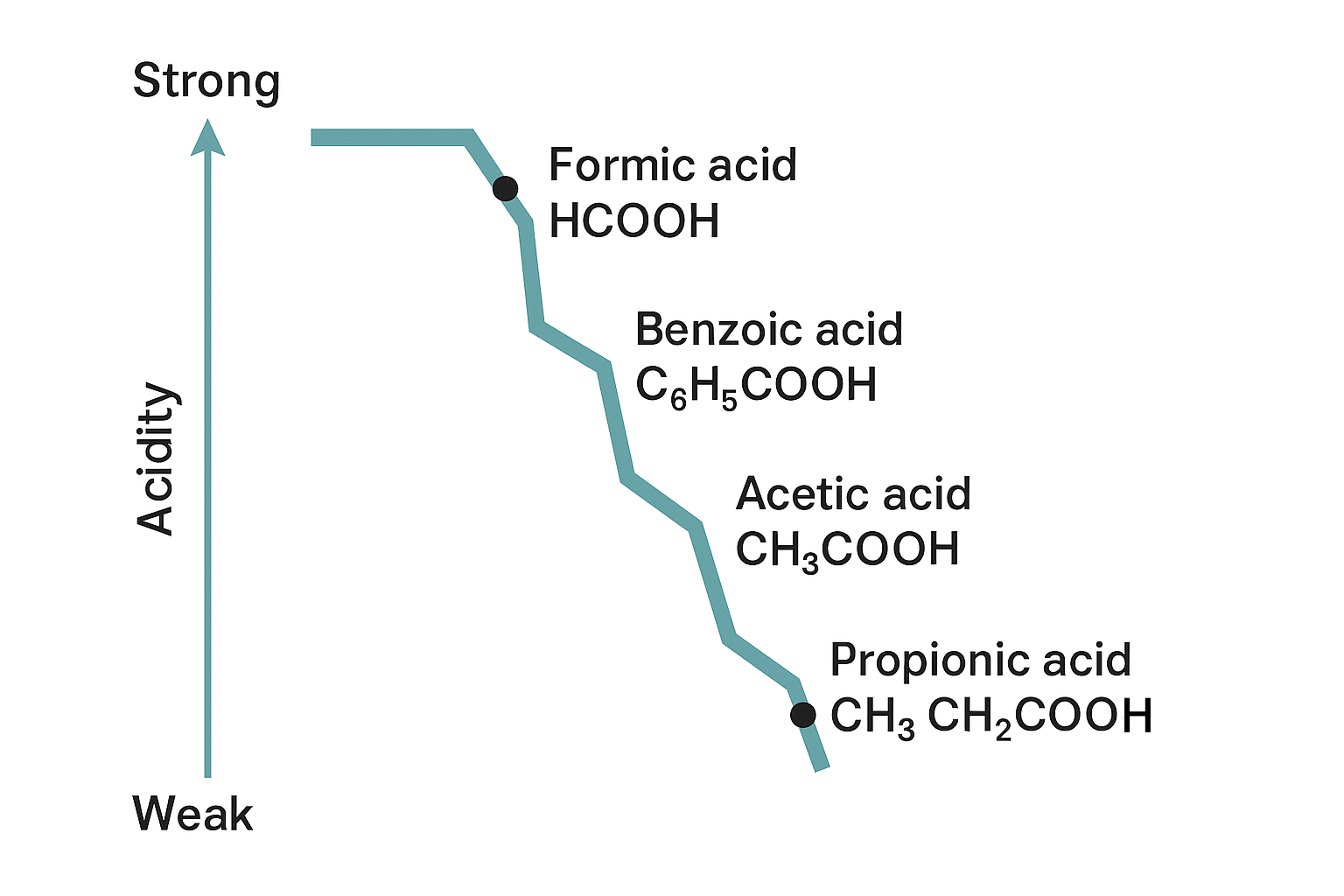
Formic Acid vs. Other Carboxylic Acids: A Relative Strength Analysis
We select acetic acid, propionic acid, and benzoic acid for comparison because they represent the most common and structurally diverse organic carboxylic acids. This comparison illustrates how molecular structure dictates $\text{pKa}$.
| Carboxylic Acid | Chemical Formula | Approximate $\text{pKa}$ | Relative Acidity | Application Connection |
|---|---|---|---|---|
| Formic Acid | HCOOH | 3.75 | Strongest | Stronger pKa is required for effective biocidal action in animal feed preservation. |
| Benzoic Acid | C6H5COOH | ~4.20 | Second Strongest | Its mild pKa makes it suitable for use as an antimicrobial food preservative (E210). |
| Acetic Acid | CH3COOH | ~4.76 | Weakest of the common ones | Milder acidity is why it’s the main component of vinegar, making it safe for consumption. |
| Propionic Acid | CH3CH2COOH | ~4.87 | Weakest overall | Used primarily to inhibit mold in bread and cheese, chosen for its safety profile and low volatility. |
The Reason Behind the Strength: No Electron-Donating Groups
The acidity of an acid hinges on the stability of its conjugate base (the ion formed after losing the H+.
- Formic Acid (HCOOH): The carboxyl group is attached only to a simple hydrogen atom. Crucially, there is no Electron-Donating Group (EDG). The lack of an EDG means the negative charge on the resultant formate ion (HCOO) is the most stable and dispersed, making it easiest for formic acid to release a proton.
- Acetic Acid (CH3COOH): The methyl group (CH3) acts as a slight EDG. It pushes electron density onto the carboxylate ion, which intensifies the negative charge and destabilizes the resulting acetate ion. This instability makes the proton harder to release, resulting in a higher pKa and a weaker acid.
The clear link between acid strength and application is evident: the superior pKa of formic acid allows it to perform more powerful chemical functions, like effective silage preservation, that milder organic acids cannot match.
Conclusion: Formic Acid is a Powerful Weak Acid
To conclusively answer the initial question, “Is formic acid a strong acid?”: No, it is not.
While formic acid (pKa ≈ 3.75) exhibits significantly higher acidity than common counterparts like acetic acid (pKa ≈ 4.76), making it one of the strongest organic acids, it fundamentally remains a weak acid because it only undergoes partial dissociation in an aqueous solution. This potent, yet controllable, level of acidity is what defines its critical role in industry and nature.